|
Alomone Labs
rabbit anti α2a ar polyclonal Rabbit Anti α2a Ar Polyclonal, supplied by Alomone Labs, used in various techniques. Bioz Stars score: 92/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/rabbit anti α2a ar polyclonal/product/Alomone Labs Average 92 stars, based on 1 article reviews
rabbit anti α2a ar polyclonal - by Bioz Stars,
2026-03
92/100 stars
|
Buy from Supplier |
|
Bioss
rabbit polyclonal anti α2a adrenergic receptor Rabbit Polyclonal Anti α2a Adrenergic Receptor, supplied by Bioss, used in various techniques. Bioz Stars score: 93/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/rabbit polyclonal anti α2a adrenergic receptor/product/Bioss Average 93 stars, based on 1 article reviews
rabbit polyclonal anti α2a adrenergic receptor - by Bioz Stars,
2026-03
93/100 stars
|
Buy from Supplier |
|
Novus Biologicals
primary antibody against rabbit anti-α2a-ar Primary Antibody Against Rabbit Anti α2a Ar, supplied by Novus Biologicals, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/primary antibody against rabbit anti-α2a-ar/product/Novus Biologicals Average 90 stars, based on 1 article reviews
primary antibody against rabbit anti-α2a-ar - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Thermo Fisher
rabbit anti-α2a-adrenoceptor Rabbit Anti α2a Adrenoceptor, supplied by Thermo Fisher, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/rabbit anti-α2a-adrenoceptor/product/Thermo Fisher Average 90 stars, based on 1 article reviews
rabbit anti-α2a-adrenoceptor - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Neuromics
rabbit anti α2a ar 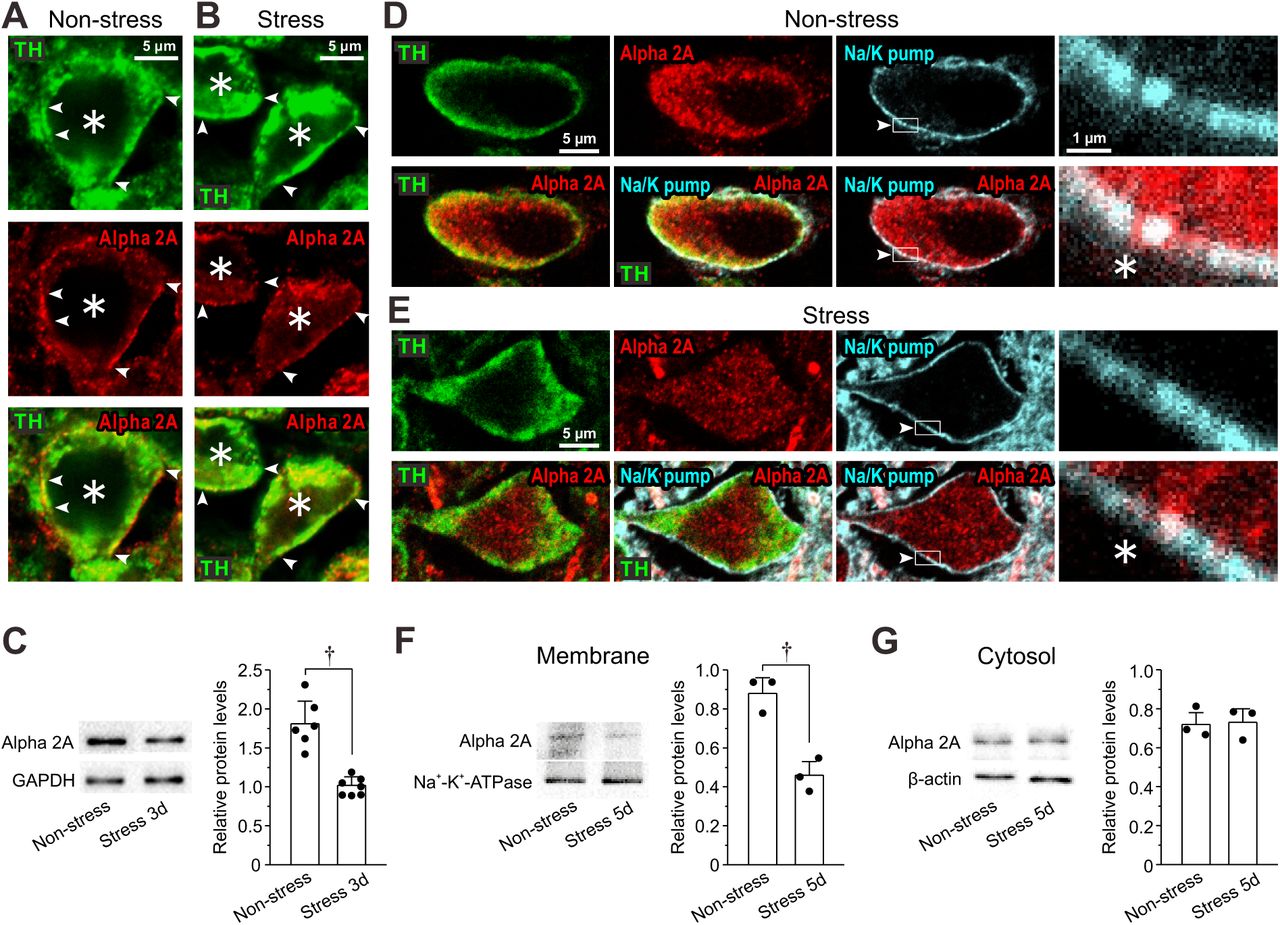 Rabbit Anti α2a Ar, supplied by Neuromics, used in various techniques. Bioz Stars score: 94/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/rabbit anti α2a ar/product/Neuromics Average 94 stars, based on 1 article reviews
rabbit anti α2a ar - by Bioz Stars,
2026-03
94/100 stars
|
Buy from Supplier |
|
Millipore
rabbit anti-α2a-ar  Rabbit Anti α2a Ar, supplied by Millipore, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/rabbit anti-α2a-ar/product/Millipore Average 90 stars, based on 1 article reviews
rabbit anti-α2a-ar - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Santa Cruz Biotechnology
anti-β2 receptors  Anti β2 Receptors, supplied by Santa Cruz Biotechnology, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti-β2 receptors/product/Santa Cruz Biotechnology Average 90 stars, based on 1 article reviews
anti-β2 receptors - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Millipore
adrenergic receptor α2a rabbit polyclonal  Adrenergic Receptor α2a Rabbit Polyclonal, supplied by Millipore, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/adrenergic receptor α2a rabbit polyclonal/product/Millipore Average 90 stars, based on 1 article reviews
adrenergic receptor α2a rabbit polyclonal - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Antibodies Inc
anti-nachr α2a polyclonal antibody (referred to as α2a from here on)  Anti Nachr α2a Polyclonal Antibody (Referred To As α2a From Here On), supplied by Antibodies Inc, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti-nachr α2a polyclonal antibody (referred to as α2a from here on)/product/Antibodies Inc Average 90 stars, based on 1 article reviews
anti-nachr α2a polyclonal antibody (referred to as α2a from here on) - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Proteintech
anti-α1b-ar antibody  Anti α1b Ar Antibody, supplied by Proteintech, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/anti-α1b-ar antibody/product/Proteintech Average 90 stars, based on 1 article reviews
anti-α1b-ar antibody - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Antibodies Inc
zebrafish-specific anti-nachr α2a polyclonal antibody  Zebrafish Specific Anti Nachr α2a Polyclonal Antibody, supplied by Antibodies Inc, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/zebrafish-specific anti-nachr α2a polyclonal antibody/product/Antibodies Inc Average 90 stars, based on 1 article reviews
zebrafish-specific anti-nachr α2a polyclonal antibody - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
|
Covance
mouse anti-ha-α2a-ar  Mouse Anti Ha α2a Ar, supplied by Covance, used in various techniques. Bioz Stars score: 90/100, based on 1 PubMed citations. ZERO BIAS - scores, article reviews, protocol conditions and more https://www.bioz.com/result/mouse anti-ha-α2a-ar/product/Covance Average 90 stars, based on 1 article reviews
mouse anti-ha-α2a-ar - by Bioz Stars,
2026-03
90/100 stars
|
Buy from Supplier |
Image Search Results

Journal: bioRxiv
Article Title: Chronic stress impairs autoinhibition in neurons of the locus coeruleus to increase asparagine endopeptidase activity
doi: 10.1101/2025.03.10.642335
Figure Lengend Snippet: ( A, B ) Confocal images of LC neurons showing immunoreactivities for TH and α2A-ARs, together with a merged one in non-stress and 3-day RS mice. Arrowheads indicate the membrane regions of TH-positive neurons, along which α2A-ARs were differentially expressed between the control and the RS mice. Asterisks indicate LC neurons. ( C ) Western blotting analyses showing the expression of α2A-ARs in non-stress and 3-day RS mice ( n = 6 and 7, respectively). Unpaired t -test, † p < 0.001. ( D, E ) Confocal images of LC neurons in non-stress and RS mice. Upper panels from left to right showing the respective immunoreactivity for TH, α2A-ARs (with rabbit anti-α2A-AR) and Na + -K + -pump and an enlarged image of the region enclosed with a rectangle (arrowheads) in its immediate left panel. Lower panels from left to right showing the merged image of TH and α2A-ARs, that of TH, α2A-ARs and Na + -K + -pump, that of α2A-ARs and Na + -K + -pump and an enlarged image of the region enclosed with a rectangle in its immediate left panel. ( F, G ) Western blotting analyses showing expressions of α2A-ARs and Na + -K + -pump in membrane fraction and those of α2A-ARs and β-actin in cytosol fraction in non-stress and 5-day RS mice ( n = 3 and 3 samples, respectively). Each sample represents the analysis result in the LC tissues obtained from 2-3 mice. Membrane; unpaired t -test, † p = 0.016. Cytosol; unpaired t -test, † p = 0.083.
Article Snippet: To examine the distribution of α2A-AR in LC neurons, mouse anti-TH (1:500; sc-25269, Santa Cruz Biotechnology, Santa Cruz, CA, USA), rabbit anti-sodium potassium ATPase (1:1000; ab76020, Abcam, Cambridge, MA, USA) and goat anti-α2A-AR (1:200; ab45871, Abcam) or
Techniques: Membrane, Control, Western Blot, Expressing
Journal: bioRxiv
Article Title: Chronic stress impairs autoinhibition in neurons of the locus coeruleus to increase asparagine endopeptidase activity
doi: 10.1101/2025.03.10.642335
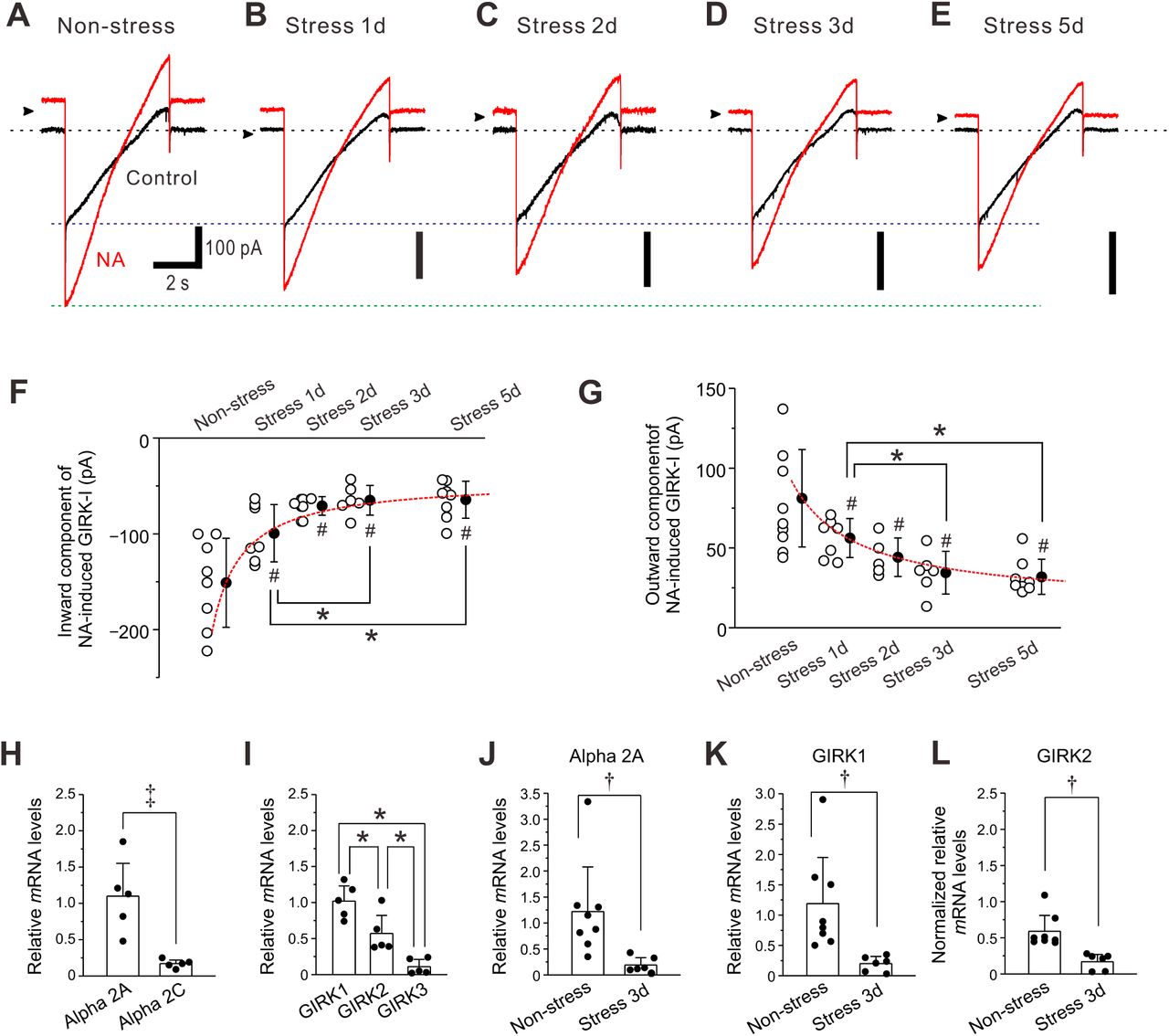
Figure Lengend Snippet: ( A-E ) Representative traces of NA-induced GIRK-I obtained from LC neurons in non-stress and 1-day, 2-day, 3-day, and 5-day RS mice. ( F, G ) Amplitudes of inward and outward components of NA-induced GIRK-I obtained from LC neurons in non-stress and 1-day, 2-day, 3-day, and 5-day RS mice ( n = 8, 7, 5, 6 and 8, respectively) decreased with the increase in the period of RS, in a way that can be described by a saturation function (red interrupted lines). The saturation level ( a + b ) and the half saturation constant ( c ) were determined by fitting the saturation function, defined as y = a + ( b * x) / ( c + x), to the data points. The values of a , b and c for the inward component of GIRK-I were −151.1, 108, and 0.9, respectively, and those for the outward component of GIRK-I were 81.1, –67, and 1.6, respectively. Inward component: one-way ANOVA, p < 0.001, post hoc fisher’s PLSD, 1-day; # p < 0.001 vs Non-stress and * p < 0.05 vs 3-day and 5-day, 2-day; # p < 0.001 vs Non-stress, 3-day; # p < 0.001 vs Non-stress, 5-day; # p < 0.001 vs Non-stress. Outward component: one-way ANOVA, p < 0.001, post hoc fisher’s PLSD, 1-day; p = 0.004 vs Non-stress and * p < 0.05 vs 3-day and 5-day, 2-day; p = 0.015 vs Non-stress, 3-day; p < 0.001 vs Non-stress, 5-day; p < 0.001 vs Non-stress. ( H ) Relative expressions of α2A and α2C mRNAs, normalized to GAPDH in LC neurons ( n = 5). Paired t -test, ‡ p = 0.014. ( I ) Relative expressions of GIRK1, GIRK2, and GIRK3 mRNAs, normalized to GAPDH in LC neurons ( n = 5). One-way RM ANOVA, p < 0.001, post hoc fisher’s PLSD; * p = 0.008 for GIRK1 vs GIRK2, * p < 0.001 for GIRK1 vs GIRK3, * p = 0.008 for GIRK2 vs GIRK3. ( J, K ) Relative expressions of α2A and GIRK1 mRNAs, respectively, normalized to GAPDH in LC neurons in non-stress mice ( n = 8), and 3-day RS mice ( n = 6). α2A-AR: unpaired t -test, † p = 0.020; GIRK1: unpaired t -test, † p = 0.013. (L) Normalized relative expressions of GIRK2 mRNA in LC neurons in non-stress mice ( n = 8) and 3-day RS mice ( n = 6), normalized to the ratio of the mean value of the relative expressions of GIRK1 mRNA to that of GIRK2 mRNA in LC neurons in non-stress mice ( I ). Unpaired t -test, † p = 0.037.
Article Snippet: To examine the distribution of α2A-AR in LC neurons, mouse anti-TH (1:500; sc-25269, Santa Cruz Biotechnology, Santa Cruz, CA, USA), rabbit anti-sodium potassium ATPase (1:1000; ab76020, Abcam, Cambridge, MA, USA) and goat anti-α2A-AR (1:200; ab45871, Abcam) or
Techniques:
![( A, B ) Differential free concentrations of NA to be metabolized by MAO-A into DOPEGAL between control and stress conditions: Under the control condition ( A ), NA in the cytosol is mostly taken up into the cytoplasmic vesicles by vMAT2 (thick arrow 3), rather than being directly metabolized by MAO-A into DOPEGAL (thin interrupted arrow). Vesicular NA is released from cell bodies as autocrine following [Ca 2+ ] i increases caused by action potentials, and the released NA activates α2A-AR-coupled GIRK channels, causing autoinhibition. Subsequently, the autocrine released NA is slowly taken up into the cytosol of LC neurons by NAT after dissociation from α2A-ARs. Under the stress condition ( B ), an activation of CRF receptors in LC neurons by stress inhibits leak K + channels and increases firing activities in LC neurons, subsequently causing a larger [Ca 2+ ] i increase together with Ca 2+ -induced Ca 2+ release (CICR). Impairment of autoinhibiton due to Ca 2+ dependent internalization of α2A-ARs-coupled GIRK channels leads to the persistent excitation in LC neurons, which enhances autocrine release of NA (thick arrow 1). Subsequently, the excessively autocrine released NA is taken up directly and rapidly by NAT into the cytosol without binding to α2A-ARs (thick arrow 2). Such a facilitation of re-uptake of NA by NAT would increase active NA storage into cytoplasmic vesicles by vMAT2 (thick arrow 3), while the rate of NA leakage from cytoplasmic vesicles would also increase (arrow 4) due to a dynamic equilibrium in cytoplasmic vesicles between active NA storage into cytoplasmic vesicles and passive NA leakage from cytoplasmic vesicles. Subsequently, such an increase in the rate of NA leakage would result in an increase in a MAO-A metabolite, DOPEGAL and AEP, leading to a production of cleaved tau N368 fragment and an impairment of learning/memory.](https://bio-rxiv-images-cdn.bioz.com/dois_ending_with_35/10__1101_slash_2025__03__10__642335/10__1101_slash_2025__03__10__642335___F6.large.jpg)
Journal: bioRxiv
Article Title: Chronic stress impairs autoinhibition in neurons of the locus coeruleus to increase asparagine endopeptidase activity
doi: 10.1101/2025.03.10.642335
Figure Lengend Snippet: ( A, B ) Differential free concentrations of NA to be metabolized by MAO-A into DOPEGAL between control and stress conditions: Under the control condition ( A ), NA in the cytosol is mostly taken up into the cytoplasmic vesicles by vMAT2 (thick arrow 3), rather than being directly metabolized by MAO-A into DOPEGAL (thin interrupted arrow). Vesicular NA is released from cell bodies as autocrine following [Ca 2+ ] i increases caused by action potentials, and the released NA activates α2A-AR-coupled GIRK channels, causing autoinhibition. Subsequently, the autocrine released NA is slowly taken up into the cytosol of LC neurons by NAT after dissociation from α2A-ARs. Under the stress condition ( B ), an activation of CRF receptors in LC neurons by stress inhibits leak K + channels and increases firing activities in LC neurons, subsequently causing a larger [Ca 2+ ] i increase together with Ca 2+ -induced Ca 2+ release (CICR). Impairment of autoinhibiton due to Ca 2+ dependent internalization of α2A-ARs-coupled GIRK channels leads to the persistent excitation in LC neurons, which enhances autocrine release of NA (thick arrow 1). Subsequently, the excessively autocrine released NA is taken up directly and rapidly by NAT into the cytosol without binding to α2A-ARs (thick arrow 2). Such a facilitation of re-uptake of NA by NAT would increase active NA storage into cytoplasmic vesicles by vMAT2 (thick arrow 3), while the rate of NA leakage from cytoplasmic vesicles would also increase (arrow 4) due to a dynamic equilibrium in cytoplasmic vesicles between active NA storage into cytoplasmic vesicles and passive NA leakage from cytoplasmic vesicles. Subsequently, such an increase in the rate of NA leakage would result in an increase in a MAO-A metabolite, DOPEGAL and AEP, leading to a production of cleaved tau N368 fragment and an impairment of learning/memory.
Article Snippet: To examine the distribution of α2A-AR in LC neurons, mouse anti-TH (1:500; sc-25269, Santa Cruz Biotechnology, Santa Cruz, CA, USA), rabbit anti-sodium potassium ATPase (1:1000; ab76020, Abcam, Cambridge, MA, USA) and goat anti-α2A-AR (1:200; ab45871, Abcam) or
Techniques: Control, Activation Assay, Binding Assay

Journal: The European journal of neuroscience
Article Title: Activation of α2A-Containing Nicotinic Acetylcholine Receptors Mediates Nicotine-Induced Motor Output in Embryonic Zebrafish
doi: 10.1111/ejn.12591
Figure Lengend Snippet: A) In situ hybridization in a 20 hpf wildtype embryo using nAChR α2A RNA probes. A photomicrograph of the head region viewed from the ventral side is shown. White arrows point to α2A nAChR mRNA expression in the region of the olfactory epithelium. Note the size of the scale bar. B) Left, same as in A, but photomicrograph at the left now shows a lateral view of spinal cord. The white dashed line separates dorsal spinal cord, where there is robust α2A nAChR mRNA expression, from ventral spinal cord where mRNA expression is weaker appearing as individual dots. Right, the mRNA expression is overlaid with anti-Hu signal in the same embryo. The large cells in dorsal spinal cord are the Rohon-Beard neurons. C) 2A nAChR subunit protein is expressed by olfactory sensory neurons as revealed by anti-α2A immuno-staining at 24 and 36 hpf. D) Left, representative images from a 22 hpf Tg(isl2b:GFP) embryo shows a dorsal view of the spinal cord region to reveal the anti-α2A immuno-staining in RB neurons. Right, merged image shows GFP positive RB neurons labeled with the α2A antibody. E) Left, photomicrograph of a 22 hpf Tg(isl2b:GFP) embryo shows GFP expression in trigeminal neurons. Middle, image shows immuno-labeling (or absence of) with the zebrafish antibody designed against the nAChR 2A subunit. The GFP positive cells in trigeminal ganglion did not label with the antibody. Right, merged image shows that immuno-staining with the α2A antibody does not label trigeminal ganglion cells. F) Top, photomicrograph shows a 30 hpf embryo labeled with the α2A antibody. White arrows point to RB neurons. Bottom, photomicrograph of a 30 hpf embryo obtained at the same exposure setting used to acquire the image in the top panel reveals immuno-staining with the α2A antibody previously incubated with the α2A peptide (α2A block peptide). The labeling of dorsal cells in spinal cord has been greatly reduced. G) Left, photomicrograph of a 33 hpf embryo reveals anti-α2A labeling in the olfactory epithelium. Right, photomicrograph of a 33 hpf embryo acquired with the same exposure setting used to acquire the image in the left panel. The embryo was incubated in the anti-α2A block peptide “cocktail”. The labeling of the olfactory epithelium is greatly reduced. White dashed circle highlights the region of the olfactory epithelium. Scale bars, 20 μm.
Article Snippet: The KLH-conjugated peptide was injected into rabbits to generate a zebrafish-specific
Techniques: In Situ Hybridization, Expressing, Immunostaining, Labeling, Immunolabeling, Incubation, Blocking Assay

Journal: The European journal of neuroscience
Article Title: Activation of α2A-Containing Nicotinic Acetylcholine Receptors Mediates Nicotine-Induced Motor Output in Embryonic Zebrafish
doi: 10.1111/ejn.12591
Figure Lengend Snippet: A) An α2A splice blocking MO was designed to target the exon2-intron2 boundary in the 6-exon nAChR-α2A transcript. The MO is predicted to result in exon 2 skipping during nAChR α2A mRNA processing in vivo. RNA from 24 and 48 hpf zebrafish were reverse-transcribed and amplified using α2A subunit specific primers. The PCR products from control MO-injected embryos and α2A morphant zebrafish predicted to amplify at ~590 bp and ~450 bp, respectively (yellow arrows point to 450 bp bands). B) Embryos (33 hpf) injected with control MO displayed nAChR α2A immunoreactivity in olfactory sensory neurons but exhibited a substantial reduction in nAChR α2A expression when injected with the α2A MO. C) Control MO-injected embryos exhibited nAChR α2A immuno-reactivity in RB neurons at 33 hpf (left, yellow arrowheads), whereas stage matched α2A morphants (middle and right) lacked α2A labeling. D) Double antibody labeling using zn12 and α2A antibodies in 31 hpf control MO-injected embryos. E) Same as is D but for 31-hpf α2A morphants. The zn12 antibody was used to label RB neurons in dorsal spinal cord to confirm the specific knockdown of the α2A subunit expression specifically in RB neurons. Dotted line in B denotes the boundary of the olfactory sensory organ. Scale bars, 20 μm.
Article Snippet: The KLH-conjugated peptide was injected into rabbits to generate a zebrafish-specific
Techniques: Blocking Assay, In Vivo, Amplification, Injection, Expressing, Labeling, Antibody Labeling

Journal: OncoTargets and therapy
Article Title: Sympathetic innervation contributes to perineural invasion of salivary adenoid cystic carcinoma via the β2-adrenergic receptor
doi: 10.2147/OTT.S190847
Figure Lengend Snippet: The primers used in the study
Article Snippet: Anti-α1B-AR and
Techniques: Sequencing

Journal: OncoTargets and therapy
Article Title: Sympathetic innervation contributes to perineural invasion of salivary adenoid cystic carcinoma via the β2-adrenergic receptor
doi: 10.2147/OTT.S190847
Figure Lengend Snippet: β2-AR was overexpressed in SACC-83 and SACC-LM cell lines. Notes: ( A , B ) qRT-PCR analysis showing α1A-AR, α1B-AR, α1D-AR, α2A-AR, α2B-AR, α2C-AR, β1-AR, β2-AR, and β3-AR mRNA expression levels in SACC-83 and SACC-LM cells. ( C ) Immunofluorescence analysis of α1B-AR, α2A-AR, β1-AR, and β2-AR expression in SACC-83 and SACC-LM cells. ( D , E ) Quantitative comparison of the mean density (IOD/area) of α1B-AR, α2A-AR, β1-AR, and β2-AR expression in SACC-83 and SACC-LM cells. Original magnification, 400×; scale bar=20 μm. Abbreviations: AR, adrenergic receptor; IOD, integrated optical density; qRT-PCR, quantitative real-time polymerase chain reaction; SACC, salivary adenoid cystic carcinoma.
Article Snippet: Anti-α1B-AR and
Techniques: Quantitative RT-PCR, Expressing, Immunofluorescence, Comparison, Real-time Polymerase Chain Reaction